The Food and Drug Administration, along with Acella Pharmaceuticals, has announced the recall of thyroid medication.
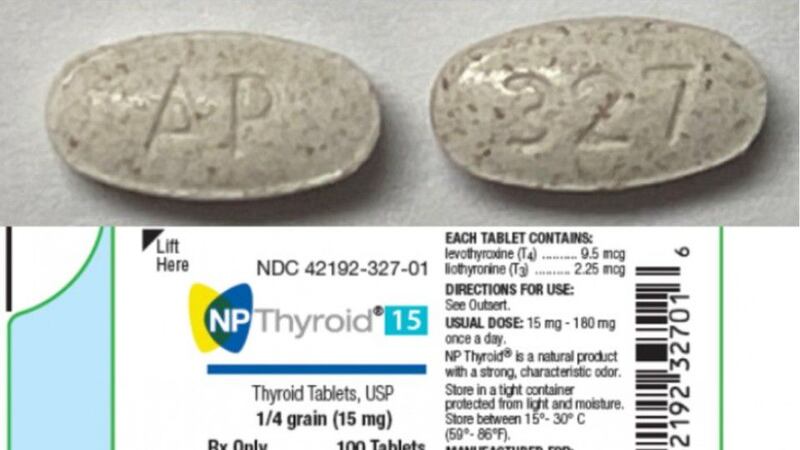
Acella Pharmaceuticals recalled one lot of 15 mg and one lot of 120 mg NP Thyroid brand tablets.
Testing discovered that the two lots may not be strong enough — as low as 87% of the labeled amount of levothyroxine, the FDA said.
The recalled 15 mg tablet has lot number M327E19-1 with an expiration date of October 2020 and NDC number of 42192-327-01.
The recalled 120 mg tablet has lot number M328F19-3 with an expiration date of November 2020 and NDC number of 42192-328-01.
If you have the recalled medication, you are being directed to contact your doctor before stopping the treatment, or if you have had any issues with taking the medication.
Consumers are being directed to call Acella Pharmaceuticals at 1-888-280-2044 or email at recall@acellapharma.com.
For more information, click here.
Cox Media Group